What is TPE
Thermoplastic elastomers (TPE) have become indispensable today. They provide the basis for greater efficiency, comfort and safety in everyday life.
What are Thermoplastic Elastomers (TPE)
TPE - Thermoplastic elastomers are an indispensable tool used by processors, product developers and designers alike. They combine the dynamic processing properties of thermoplastic plastics with the softness and flexibility of elastomers.
The different types of TPE
The different types of TPEs and their modification options provide the basis for plenty of materials properties thus enabling a cost-effective processing in applications for the most diverse industries.
TPEs contribute to enhancing products and setting them apart from others. In addition, they take over a lot of technical functions that so far have been reserved for elastomers. Using TPEs does not only increase a product’s benefits but also brings economic advantages to processors.
The processing and behavior of TPE materials classify them as belonging to a group of materials between thermoplastics and elastomers. They form an independent class of materials.
Basically, a distinction is made between reactor-made TPEs (e.g. TPA, TPU, and TPC) and TPE compounds (e.g. TPS and TPV). The properties of reactor-made TPEs are implemented in one polymer. The properties of TPE blends result from mixing different polymers to form a so-called compound.
TPS
TPS is a TPE based on styrene block copolymers (SBC). Depending on the application, KRAIBURG TPE uses various styrene block copolymers to produce TPS compounds, which differ in their property profile and are decisive for the design of the product properties. SBSs are characterized by their polymer structure and enable to create a wide variety of TPS compounds that meet diverse market requirements ranging from the automotive sector to the pharmaceutical industry.
How is TPE processed?
Like thermoplastics, TPEs are heated to process them by means of injection molding or extrusion. As they show their original elastic properties again after cooling down, they are recyclable like thermoplastics. In contrast to TPEs, elastomers cannot be processed by melting; they are chemically crosslinked and not recyclable.
The strongest arguments in favor of using TPE
- Easy thermoplastic processing
- Short cycle times
- Low energy consumption
- Multi-component processing and thus reduced assembly costs
- Combination of different materials (e.g. hard-soft composite)
- 100% recyclable in processing
- A wide processing window
- Plenty of coloring options, including color effects for more design flexibility
- Many possible applications ranging from the automotive sector to the pharmaceutical industry.
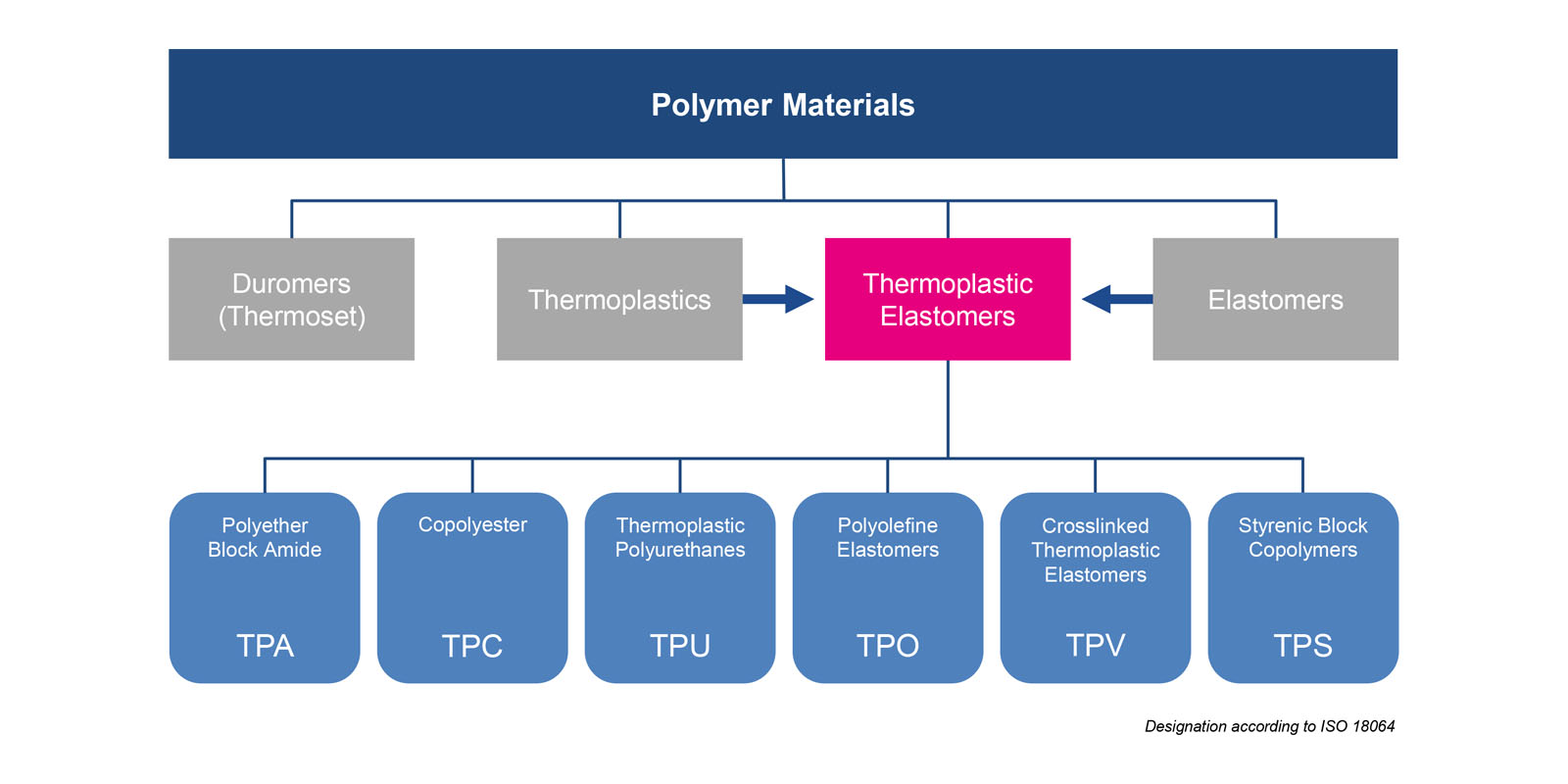
Classification
Polymers are classified into four groups: thermoplastics, thermosets, elastomers and thermoplastic elastomers (TPEs).
Thermoplastics consist of polymer chains and are highly entangled. They are kept together by interactions between molecules such as van der Waals forces. Thermal effects and shear forces allow them to be processed by injection molding. When cooled down the material solidifies again. As this process is purely physical, it can be repeated at any time (example: polypropylene).
Thermosets such as epoxy resins are polymers that are tightly crosslinked via chemical bonds and do not melt. They maintain their crosslinked structure even at high temperatures and are characterized by high strength and stiffness. Under stress, they are subject to brittle fracture (example: epoxy resins).
Elastomers, often referred to as rubbers, are chemically crosslinked polymers. They are considerably less densely crosslinked than thermosets, leaving larger elastic areas between the individual cross-links. These areas are crucial to their elastic properties. Elastomers cannot be melted again. Once they have been crosslinked they will maintain their shape. Examples of elastomers are natural rubber or synthetic rubber such as EPDM and NBR.
Basically, thermoplastic elastomers can be divided into two types: reactor-made TPEs (e.g. TPA, TPU, and TPC) and TPE compounds (e.g. TPS or TPV). The properties of reactor-made TPEs are implemented in one polymer. The properties of TPE compounds result from mixing two polymers to form a so-called compound.
What is TPE used for?
TPEs are used in a versatile range of applications in various sectors, meeting the industry-specific requirements. They are used not only for control elements in automotive interiors but also for exterior window encapsulations and under-the-hood seals. In industrial products, they are used for tool handles and cable grommets, for example. In consumer goods, TPEs are found in toys, sports equipment, packaging, and hygiene articles such as toothbrushes and razors. For medical applications, special compounds that meet the strict requirements in this sector have been developed. These applications include drip chambers, seals and medical tubing.
KRAIBURG TPE offers an application engineered portfolio for various ranges of performance and makes use of the TPE classes suitable for each application.
List of terms and definitions
TPE compounds: TPEs that are compounded using at least two different types of polymers. One polymer type adds elastic properties to the compound, the other one provides thermoplastic properties (this group includes TPV, TPS, and TEH compounds).
SBC: Styrene block copolymer
TPS: Thermoplastic styrene block copolymer
Various SBC types:
- SBS: Styrene-butadiene-styrene block copolymer
- SIS: Styrene-isoprene-styrene block copolymer
- SEBS: Styrene-ethylene-butylene-styrene block copolymer
- SEEPS: Styrene-ethylene-ethylene-propylene-styrene block-copolymer
- SEPS: Styrene-ethylene-propylene-styrene block copolymer
- SEPS-V: Styrene-ethylene-propylene-styrene block copolymer, cross-linkable
TPV: Thermoplastic elastomer of thermoplastic and vulcanized elastomers (e.g. EPDM/ PP)
TEH: Thermoplastic elastomer hybrid (alternatives to crosslinked rubber that can be processed by efficient thermoplastic processing)
TPO: Thermoplastic elsatomer polyolefin (both as a reactor-made TPE and a TPE blend
Reactor-made TPE: The TPE material properties are provided by a single polymer type. The TPE is created immediately in the polymerization process. The well-known reactor-made TPEs are obtained by copolymerization of at least two monomers, which are block polymerized.
TPU: Thermoplastic polyurethane elastomer
TPC: Thermoplastic copolyester elastomer
TPA: Thermoplastic polyether block amides
TPO: Thermoplastic polyolefin (both as a reactor-made TPE and a TPE blend)
Thermoplastic Polyurethane (TPU)
Thermoplastic polyester elastomers (TPC)
Thermoplastic vulcanizates (TPV)
Thermoplastic polyamide elastomers (TPA)
Thermoplastic polyolefins (TPO)
Thermoplastic styrene block copolymers (TPS)